This web page was produced as an assignment for Genetics 677 an undergraduate course at UW-Madison
What are protein motifs and domains?
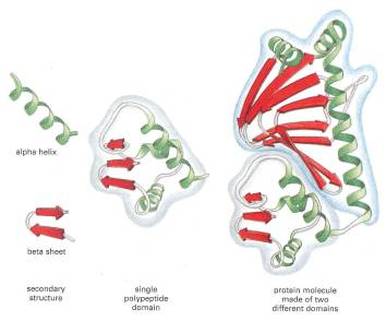
Fig 1: This particular protein is made up of two different domains.
Motifs and domains are units within the protein themselves. They are especially important in classifying protein families. Contrary to popular belief, motifs and domains are not the same. Motifs are used to describe what is known as the supersecondary structure of the protein [1]. This is the combination of alpha-helices and beta-structures in the protein-the combination of secondary structural elements [1] Essentially, the supersecondary structure describes the folding patterns of the secondary structure of the protein and how those secondary structures interact. However, motifs are used to describe the structure of a protein only. They cannot predict function of a protein. Domains, however, are seen as functional units of a protein [2]. Domains are responsible for a specific function or interaction of a protein [2]. Domains are a sequence of amino acids that are conserved between many different organisms. They are often used to hypothesize the overall function of uncharacterized proteins because the sequence of amino acids usually carries a similar function. However, domains can be found in proteins that function differently [2] Overall, domains are units within the protein that are essential for the proteins function and are an important part to take into account when analyzing a protein.
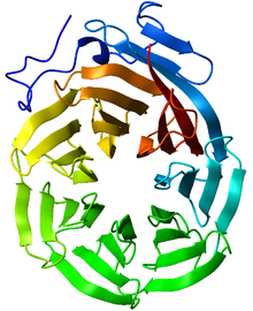
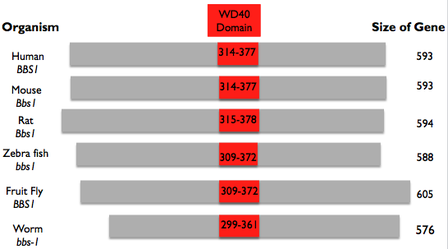
The WD40 Domain and BBS1Before 2003, no domains had been identified in the BBS1 protein. Its function and purpose in the primary cilia was mainly unknown. In 2003, however, a paper was published that predicted a β-propeller motif in BBS1, BBS2, and BBS7 [3]. It was later confirmed by other scientist that this motif was in fact present in these genes. Using the paper that was published in 2003 and ClustalOmega, I aligned the sequences for all the model organisms up and found the β-propeller region in all of them. The results of this are shown on the right in Fig. 3. This domain gave insight into the function of these BBS genes and provided insight in to the function of the BBSome.
The β-propeller motif is formed through the WD40 domain. The WD40 domain consists of WD40 repeats, which are 40 amino acid stretches in the protein [4]. Tandem copies of this repeat cause the protein to fold in a circular solenoid domain, which is how it is known as the WD40 domain [4] This circular folding of the protein forms a β-propeller structure. This structure is implicated in a variety of functions including signal transduction, transcription regulation, and cell cycle control [4]. The underlying common function of this domain, however, is coordinating multi-protein complex assemblies [4]. It serves as a rigid scaffold for these protein interactions [4]. This domain was highly conserved throughout BBS1 genes. One thing to note is that the most common mutation in the BBS1 gene that causes Bardet-Biedl syndrome, the M390R mutation, is outside of this domain. Therefore, this mutation does not change this domain in the protein. |
|
Analysis and Discussion
The WD40 is particularly interesting and insightful into the understanding of the BBS1 protein. The main function of the WD40 domain is coordinating the assembly of mutli-protein complexes such as the BBSome [4]. This domain is also found in two other BBS genes that are a part of the BBSome-BBS2 and BBS7 [3] This domain indicates it plays a role in the assembly of the BBSome and also that BBS1 is an important player in the formation of this complex. Furthermore, it is found in BBS1 genes in other organisms indicating that this is a very important domain in the function of this protein. All of this indicates that the BBS1 gene plays a very important role in coordinating the assembly of the BBSome and providing a backbone for this protein complex. It serves as an important protein in that it forms the functioning unit-the BBSome. However, the M390R mutation that causes Bardet-Biedl Syndrome falls outside of this domain. Further studies should be done to see if there are other domains within this protein.
References:
[Banner Photo] "NDM-1" NDM-1. Retrieved 12 March 2013 from http://www.personal.psu.edu/czc5161/blogs/testing/references.html.
[1] “Introduction to Protein Classification at the EBI.” The European Bioinformatics Institute. Retrieved 7 April 2013 from http://www.ebi.ac.uk/training/online/course/introduction-protein-classification-ebi/protein-classification/what-are-protein-domains
[2] “Supersecondary Structures (Motifs) and Domains.” The Biochemistry Questions Site: A free Biochemistry Question Bank for premed, medical students and FMG. Retrieved 7 April 2013 from http://biochemistryquestions.wordpress.com/2008/10/09/supersecondary-structures-motifs-and-domains/
[3] Jose´ L. Badano, Stephen J. Ansley, Carmen C. Leitch, Richard Alan Lewis, James R. Lupski, and Nicholas Katsanis. (2007). Identification of a Novel Bardet-Biedl Syndrome Protein, BBS7, That Shares Structural Features with BBS1 and BBS2. American Journal of Human Genetics, 72(3), 650-8.
[4] “Family: WD40 (PF00400).” Wellcome Trust Sanger institute. Retrieved 8 April 2013 from http://pfam.sanger.ac.uk/family/PF00400.
[Banner Photo] "NDM-1" NDM-1. Retrieved 12 March 2013 from http://www.personal.psu.edu/czc5161/blogs/testing/references.html.
[1] “Introduction to Protein Classification at the EBI.” The European Bioinformatics Institute. Retrieved 7 April 2013 from http://www.ebi.ac.uk/training/online/course/introduction-protein-classification-ebi/protein-classification/what-are-protein-domains
[2] “Supersecondary Structures (Motifs) and Domains.” The Biochemistry Questions Site: A free Biochemistry Question Bank for premed, medical students and FMG. Retrieved 7 April 2013 from http://biochemistryquestions.wordpress.com/2008/10/09/supersecondary-structures-motifs-and-domains/
[3] Jose´ L. Badano, Stephen J. Ansley, Carmen C. Leitch, Richard Alan Lewis, James R. Lupski, and Nicholas Katsanis. (2007). Identification of a Novel Bardet-Biedl Syndrome Protein, BBS7, That Shares Structural Features with BBS1 and BBS2. American Journal of Human Genetics, 72(3), 650-8.
[4] “Family: WD40 (PF00400).” Wellcome Trust Sanger institute. Retrieved 8 April 2013 from http://pfam.sanger.ac.uk/family/PF00400.