This web page was produced as an assignment for Genetics 677 an undergraduate course at UW-Madison
Obesity and Bardet-Biedl Syndrome
Fig 1: Obesity is a common symptom of Bardet-Biedl Syndrome.
Bardet-Biedl Syndrome is characterized by a wide range of symptoms. One of these symptoms is obesity. Obesity is one the most common symptoms of Bardet-Biedl Syndrome and occurs in a large percentage of those affected by this disease. It increases a persons risk for other diseases related to obesity such as type II diabetes, heart problems, and more. Currently, obesity is thought to occur in those with Bardet-Biedl syndrome due to mistrafficking of the leptin receptor to the primary cilia [1]. Leptin is the hormone that regulates energy intake and expenditure through regulating both appetite and hunger [1]. By mistrafficking the leptin receptor to the primary cilia, Bardet-Biedl patients do not properly sense the amount of energy they have taken in and therefore eat more to obtain the energy they think they need. This causes patients to put on a lot of extra weight.
One of the problems with this current understanding is that fat accumulation is known to be controlled by many different genes and pathways [2]. This is just one pathway that describes this symptom. The probability that there is another pathway that is affected that could contribute to greater fat accumulation is very high.
One of the problems with this current understanding is that fat accumulation is known to be controlled by many different genes and pathways [2]. This is just one pathway that describes this symptom. The probability that there is another pathway that is affected that could contribute to greater fat accumulation is very high.
Proposed Experiment
C. elegans and Fat AccumulationUnlike humans, C. elegans lack leptin [2]. Therefore, their fat accumulation cannot be due to a lack of the receptor since the hormone is not even part of their fat regulatory system. Despite the lack of leptin, C. elegans do have a similar fat regulatory system as humans [2]. They have commonly been used to study the fat regulation system in humans and it has been found that many hormones, such as insulin, are conserved between C. elegans and humans [2]. So one would question what could be causing this greater fat accumulation in C. elegans. By studying this, a new understanding of how more fat is accumulated into those affected by this disease could be revealed. This organism will be a good model organism of my proposed study of looking into another pathway that could be causing greater fat accumulation.
|
What Proteins that Interact with BBS1 are Involved in Fat Regulation?
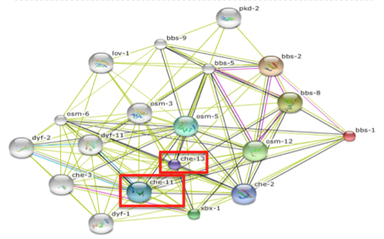
Fig 3: This is the protein network of BBS1 found in C. elegans.
The first thing to look at was to see what proteins interacted with bbs-1 in C. elegans that could lead to this phenotype. The protein network was obtained through STRING and all the proteins and their functions were analyzed. Two proteins on the network stood out: che-11 and che-13. After researching these molecules on wormbase, it was found that these proteins traffic molecules through the cilium in the intraflagellar transport system [3]. Specifically, they were said to function in formation of sensory cilia [3]. The final important aspect was that these proteins had an ortholog and homolog in humans. For che-11, its ortholog is KIAA0590 and for che-13 the homolog is IFT57/Hippi [3]. These functions led to the idea that there could be a defect in the sensing of the internal homeostasis of fat.
Looking at che-11 and che-13The first step to do is to confirm that the che-11 and che-13 proteins have a necessary role in fat accumulation. The way in which to do this would be to perform an RNAi in C. elegans and confirm that when those proteins are not present, that there is greater fat accumulation.
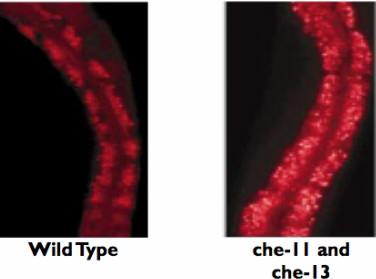
By doing and RNAi screen and using Niles Red to visually see the fat accumulation, one would expect that when the proteins are not there that there would be a greater fat accumulation. The expected results from this experiment can be seen on the right. As one can see, there are many more red fat droplets in the che-11 and che-13 mutants versus the normal wild type. This result would confirm that che-11 and che-13 have a necessary role in fat accumulation because when the proteins are absent, the organism accumulates more fat. |
What Pathway Could Be Causing the Greater Fat Accumulation?
Even though che-11 and che-13 may have a necessary role in fat regulation, how this is done is also unknown. Figuring how che-11 and che-13 function in this pathway would be the next step in the experiment. Although unknown, one hypothesis of their role in this pathway has to do with a necessary role in the sensing of insulin in the C. elegans. Insulin is the hormone that regulates carbohydrate and fat metabolism in the body. This would indicate that their homologs in humans have the same function. This hypothesis formed from a few pieces of evidence.
One piece of evidence is that one of the phenotypes associated with che-11 and che-13 mutants is a defective, smaller, dauer formation [3]. Dauer is when there is a lack of nutrients for the worm and therefore the worm does not produce insulin [4]. The lack of insulin in the worm causes a dauer formation [4]. This formation allows the worm to shut down necessary body functions to survive [4]. It allows the worm to be revived later and live a normal life [4]. The ability to enter into this state is defected in che-11 and che-13 mutants.
The other piece of evidence is that Bardet-Biedl patients are at a higher risk for Type II diabetes [5]. This is where the body does not produce enough insulin or stops making it all together later on in life unlike Type I diabetes. People who tend to get this type are usually overweight or obese. It is thought to occur because the body is creating so much insulin it eventually becomes resistant to the hormone and has to create more and more for the same response to the hormone. The body eventually becomes resistant to the hormone and then does not make it.
These two pieces of evidence coupled with the fact that che-11 and che-13 mutants cause a greater accumulation of fat indicates that there could be a greater release of insulin in C. elegans with Bardet-Biedl syndrome. This itself could be due to the fact that the sensory neurons are defective and are unable to sense the homeostasis of fat in the body and cause a greater amount to be released than normal. This can be investigated by seeing the amount of insulin released in the C. elegans in che-11, che-13, and bbs-1 mutants.
One piece of evidence is that one of the phenotypes associated with che-11 and che-13 mutants is a defective, smaller, dauer formation [3]. Dauer is when there is a lack of nutrients for the worm and therefore the worm does not produce insulin [4]. The lack of insulin in the worm causes a dauer formation [4]. This formation allows the worm to shut down necessary body functions to survive [4]. It allows the worm to be revived later and live a normal life [4]. The ability to enter into this state is defected in che-11 and che-13 mutants.
The other piece of evidence is that Bardet-Biedl patients are at a higher risk for Type II diabetes [5]. This is where the body does not produce enough insulin or stops making it all together later on in life unlike Type I diabetes. People who tend to get this type are usually overweight or obese. It is thought to occur because the body is creating so much insulin it eventually becomes resistant to the hormone and has to create more and more for the same response to the hormone. The body eventually becomes resistant to the hormone and then does not make it.
These two pieces of evidence coupled with the fact that che-11 and che-13 mutants cause a greater accumulation of fat indicates that there could be a greater release of insulin in C. elegans with Bardet-Biedl syndrome. This itself could be due to the fact that the sensory neurons are defective and are unable to sense the homeostasis of fat in the body and cause a greater amount to be released than normal. This can be investigated by seeing the amount of insulin released in the C. elegans in che-11, che-13, and bbs-1 mutants.
Looking at che-11 and che-13's role in Insulin PathwayOne way that this can be looked at is through a microarray of the insulin-like genes. C. elegans have 38 insulin like genes [2] and a microarray is an easy way to look at all of these genes at once in the different mutants. Comparing the gene expression in neural tissue between che-11, che-13, and bbs-1 to the wild-type will show if these mutants express more insulin than normal. If they do, this shows that in che-11, che-13, and bbs-1 mutants cannot sense the amount of fat in the body and therefore produce to much.
|
Why is this Important?
This experiment is proposed for the purpose of better understanding how obesity is caused in those affected with this disease. Bardet-Biedl patients suffer from a wide array of symptoms. Obesity is one of them and it can lead to many other diseases since it is a high risk factor for things such as heart disease, joints issues, and more. If one can fully understand what is being affected in the fat regulatory pathway in Bardet-Biedl syndrome, one can better treat this symptom and better help those with this disease.
The final presentation for this class can be downloaded below.
The final presentation for this class can be downloaded below.
Final Presentation For Genetics 677
| storyboard.pptx | |
| File Size: | 4171 kb |
| File Type: | pptx |
References:
[Banner Photo]"NDM-1" NDM-1. Retrieved 12 March 2013 from http://www.personal.psu.edu/czc5161/blogs/testing/references.html.
[1] Seo, S. et al. (2009). Requirement of Bardet-Biedl syndrome proteins for leptin receptor signaling. Human Moelcular Genetics, 18(7). doi: 10.1093/hmg/ddp031.[2] Mak, H. Y. et al. (2006).
[2] Polygenic control of Caenorhabditis elegans fat storage. Nature Genetics 38. doi: 10.1038/ng1739.
[3] "Wormbase." Retrieved 6 May 2013 from http://www.wormbase.org/#01-23-6.
[4] Fielenbach N and Antebi A. C. elegans dauer formation and the molecular basis of plasticity. Genes and Development, 22 (16), 2149-65. doi: 10.1101/gad.1701508.
[5] "BBS1." Genetic Home Reference: Your Guide to Understanding Genetic Conditions. Retrieved 7 Feb 2013 from http://ghr.nlm.nih.gov/gene/BBS1
[Banner Photo]"NDM-1" NDM-1. Retrieved 12 March 2013 from http://www.personal.psu.edu/czc5161/blogs/testing/references.html.
[1] Seo, S. et al. (2009). Requirement of Bardet-Biedl syndrome proteins for leptin receptor signaling. Human Moelcular Genetics, 18(7). doi: 10.1093/hmg/ddp031.[2] Mak, H. Y. et al. (2006).
[2] Polygenic control of Caenorhabditis elegans fat storage. Nature Genetics 38. doi: 10.1038/ng1739.
[3] "Wormbase." Retrieved 6 May 2013 from http://www.wormbase.org/#01-23-6.
[4] Fielenbach N and Antebi A. C. elegans dauer formation and the molecular basis of plasticity. Genes and Development, 22 (16), 2149-65. doi: 10.1101/gad.1701508.
[5] "BBS1." Genetic Home Reference: Your Guide to Understanding Genetic Conditions. Retrieved 7 Feb 2013 from http://ghr.nlm.nih.gov/gene/BBS1